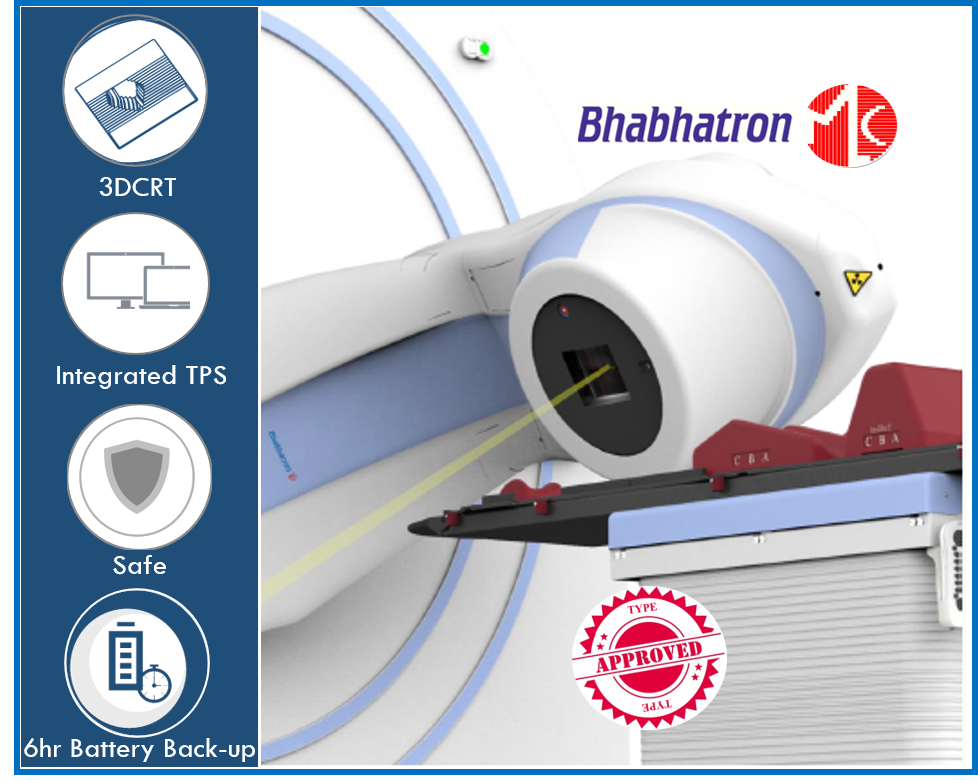
Panacea Medical Technologies Pvt. Ltd. the leading manufacturer of Radiotherapy and Radiology Devices head quartered in Malur, India has received Type Approval Certificate from Atomic Energy Regulatory Board, India for its Bhabhatron MLC Cobalt Therapy Unit.
Bhabhatron MLC technology was transferred from Bhabha Atomic Research Centre (BARC), India to Panacea Medical Technologies. Bhabhatron MLC marries the robustness of cobalt therapy with multi-leaf collimator enabling sophisticated newer 3D conformal radiotherapy techniques for enhanced clinical outcome. Bhabhatron MLC is the first Cobalt therapy unit in India which can deliver 3D CRT. Its 6-hours battery back-up together with Treatment Planning System (TPS) is especially designed and integrated to assist remote clinics treat patients unhindered by problems of power outages and practice newer precision-based radiotherapy techniques even in adverse circumstances.
This feat was possible because of the unrelenting faith and support posed by the customers, unwavering support of BARC and the regulatory board for its constant review and feedback. Bhabhatron MLC solution will be able to match and meet the demand of the Indian radiotherapy fraternity in serving the patient with quality assured 3D conformal radiotherapy needs.
Panacea Medical Technologies is an ISO-13485:2016 certified medical equipment manufacturer, pioneering state-of-art innovations and solutions for the diagnosis and treatment of cancer.
We are a technology-based company with a team of proficient, purposeful, and motivated engineers, with expertise in advanced medical equipment design, constantly at work to develop exemplary products. Our Research and Development centre is supported by the state-of-the-art manufacturing facility.

Congratulations!